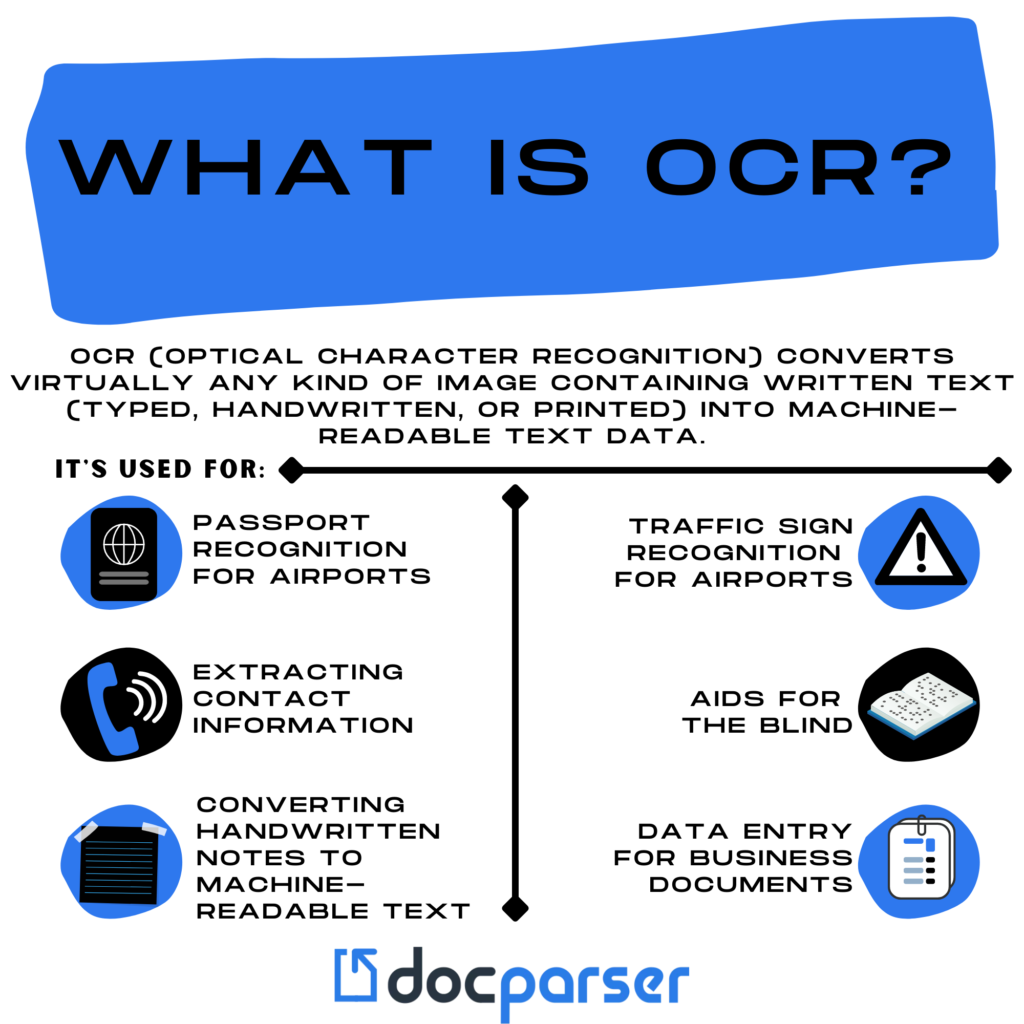
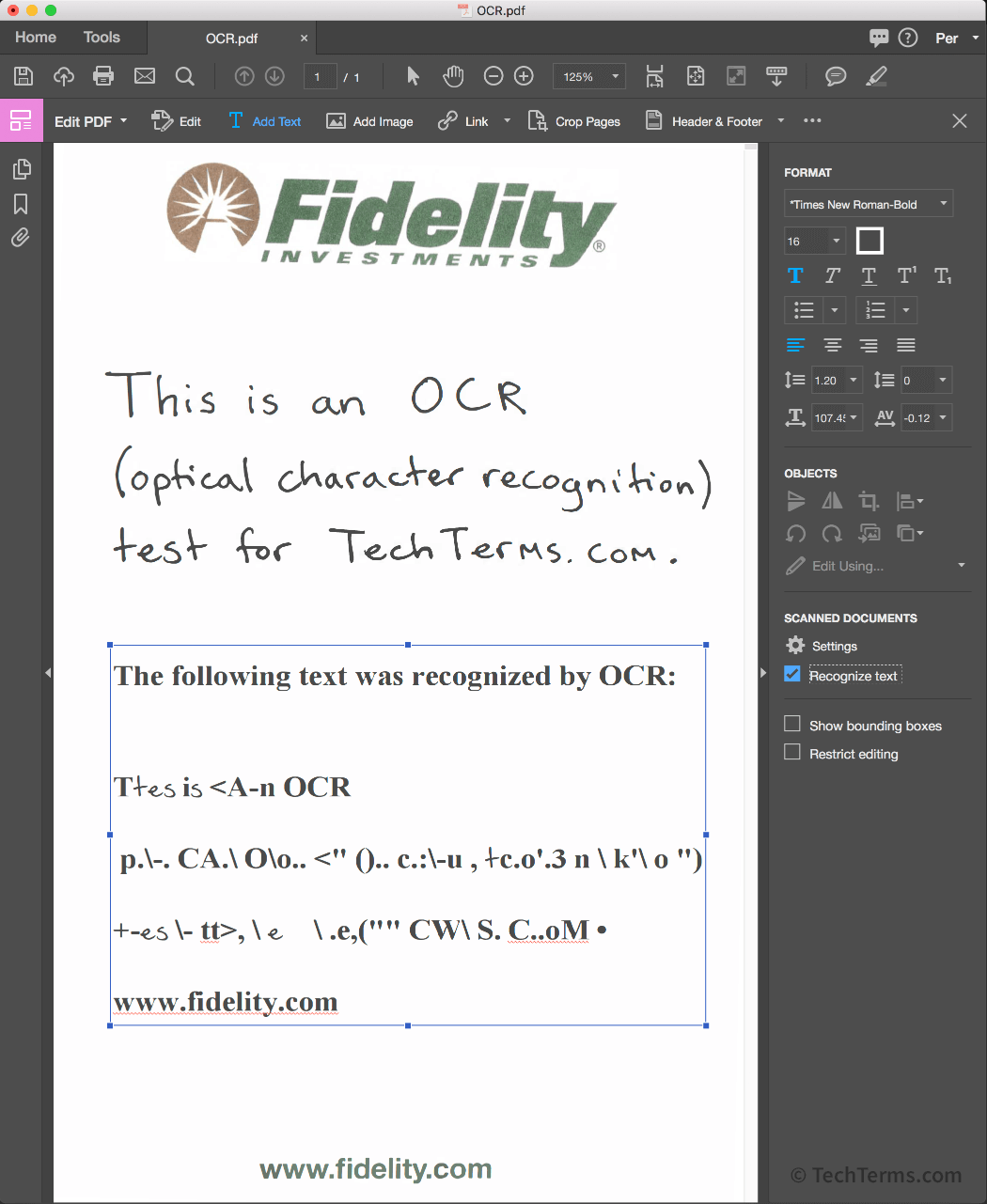
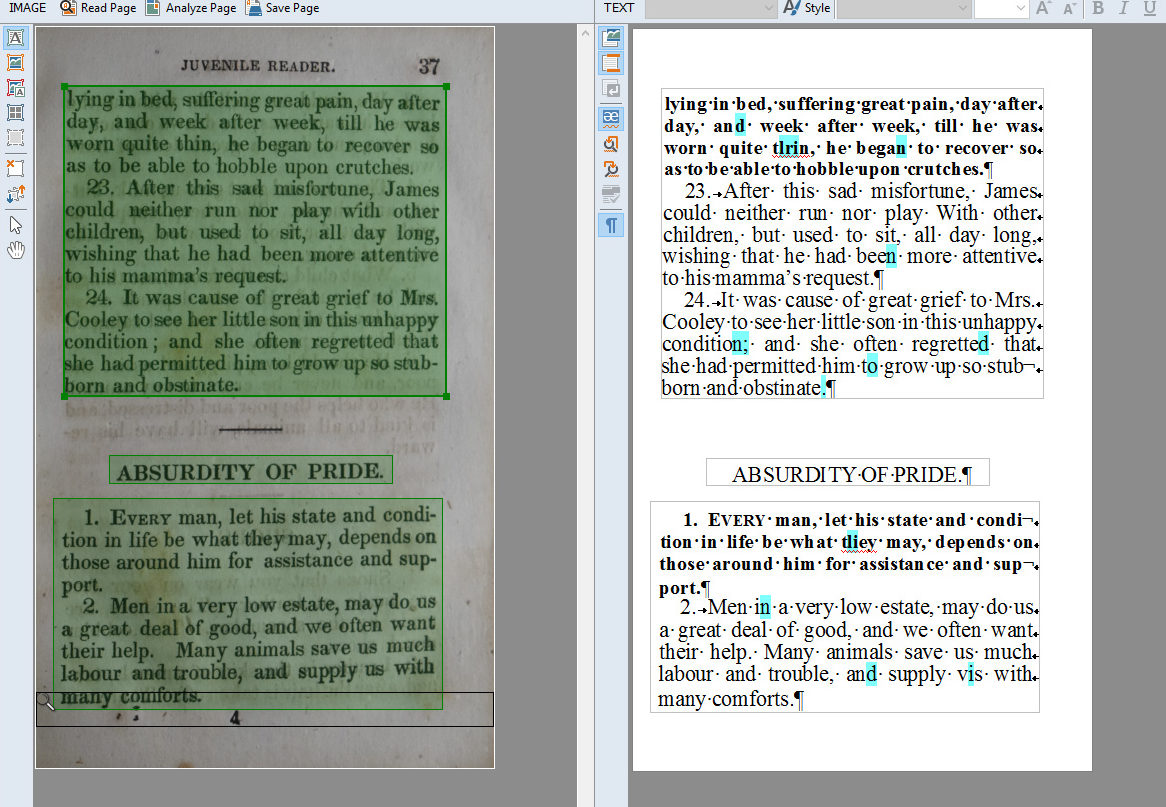
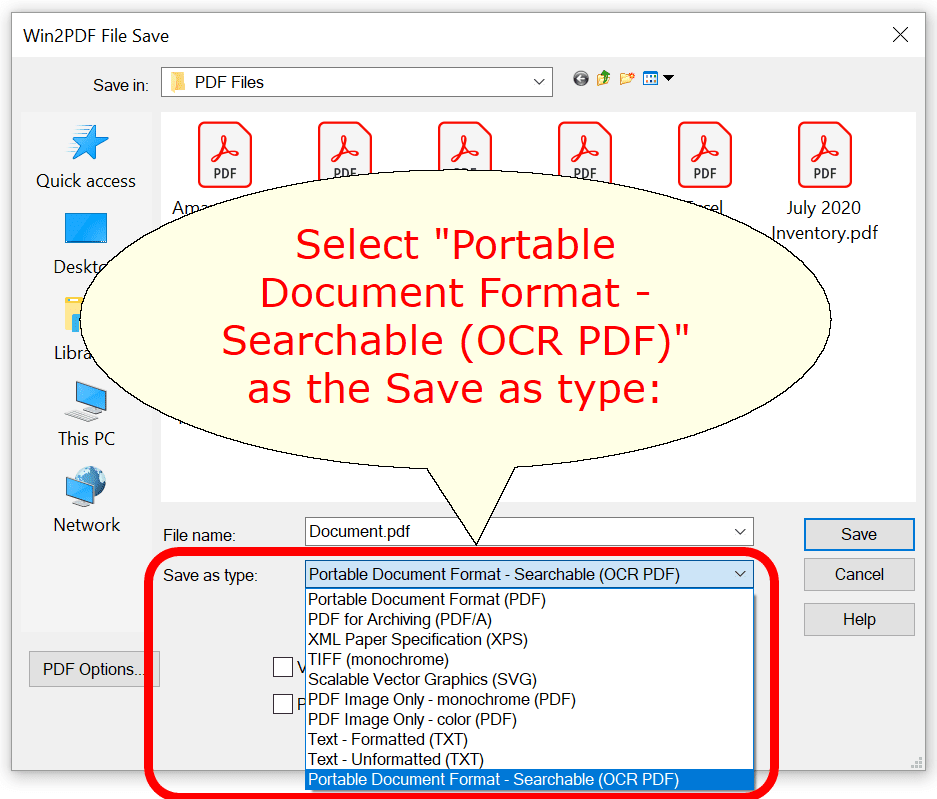
Amazon.com: Scanmarker Air Pen Scanner - OCR Digital Highlighter and Reader - Wireless (Black, Scanmarker Air) : Office Products

2023 Honda CR-V EX-L w/o BSI 4dr All-Wheel Drive Crossover: Trim Details, Reviews, Prices, Specs, Photos and Incentives | Autoblog
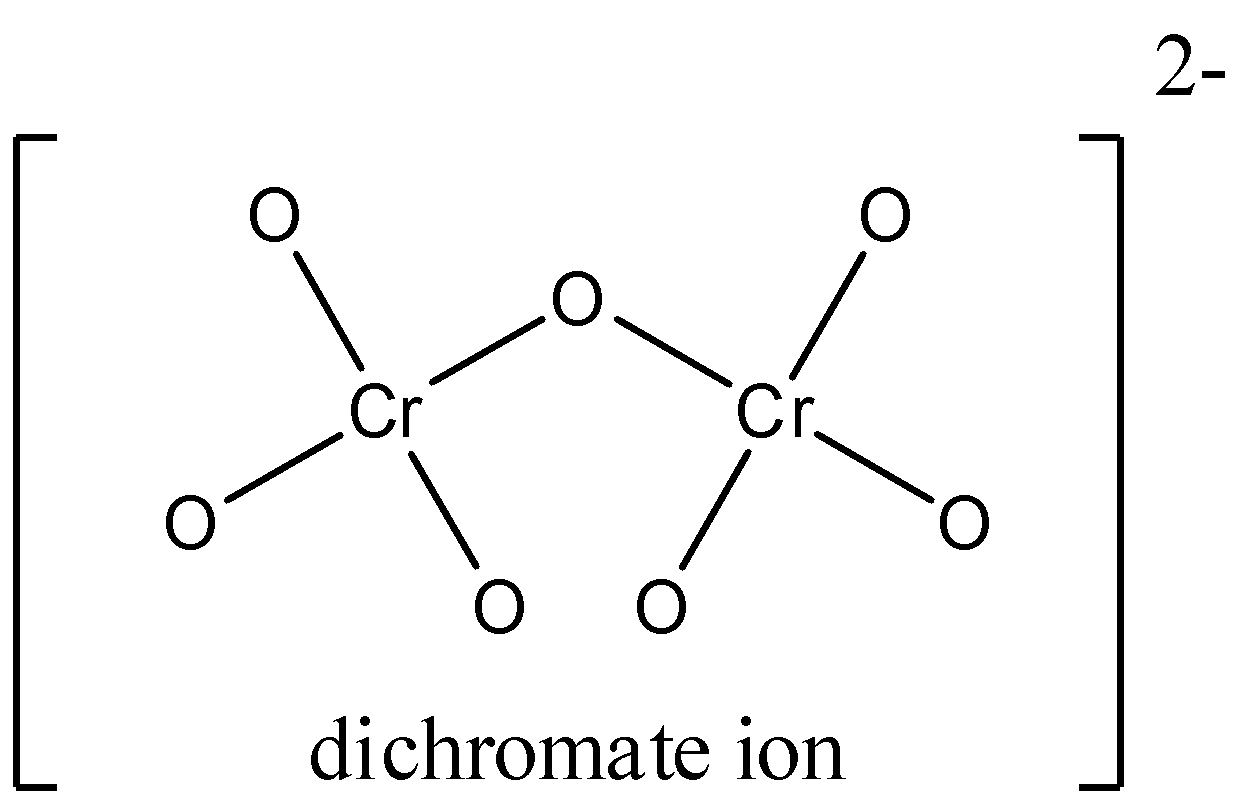
Number of $Cr - O$ bonds in dichromate ion $\\left( {C{r_2}{O_7}^{2 - }} \\right)$ is.A) $6$ B) $7$C) $8$D) $4$
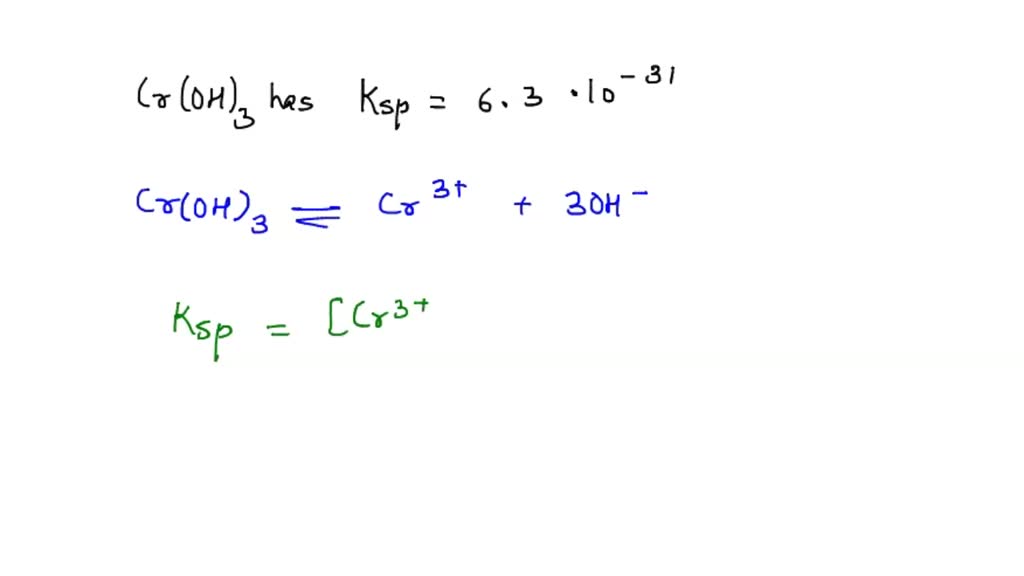
SOLVED: Chromium(III) hydroxide, Cr(OH)3, is highly water insoluble. Its molar mass is 103.02 g/mol. a.) Write the chemical equation for dissolving Cr(OH)3 in water. b.) Calculate the mass solubility of Cr(OH)3 in

Water Decontamination from Cr(III)–Organic Complexes Based on Pyrite/H2O2: Performance, Mechanism, and Validation | Environmental Science & Technology

Metal–Organic Framework MIL-101(Cr) as an Efficient Heterogeneous Catalyst for Clean Synthesis of Benzoazoles | ACS Omega
Hydrogen adsorption on two catalysts for the ortho- to parahydrogen conversion: Cr-doped silica and ferric oxide gel